Smith & Nephew — Human Factors
Improving safety and usability in a prototype abdominal closure system through human factors research
Identified usability risks and design breakdowns preventing safe and effective use in simulated surgical environments — generating evidence-based findings to inform safer concept iteration before further development investment.
Context & Constraints
A prototype abdominal closure system presented usability challenges that risked incorrect use in surgical contexts. Users included surgeons with widely varying techniques, experience levels, and environmental pressures — making it impossible to rely on a single mental model or usage pattern. The primary constraint was evaluating and improving usability without compromising clinical realism, safety protocols, or regulatory expectations. Findings needed to be defensible and actionable enough to inform the next phase of development — and precise enough to distinguish between issues of concept design, interaction design, and procedural communication.
Key Insights
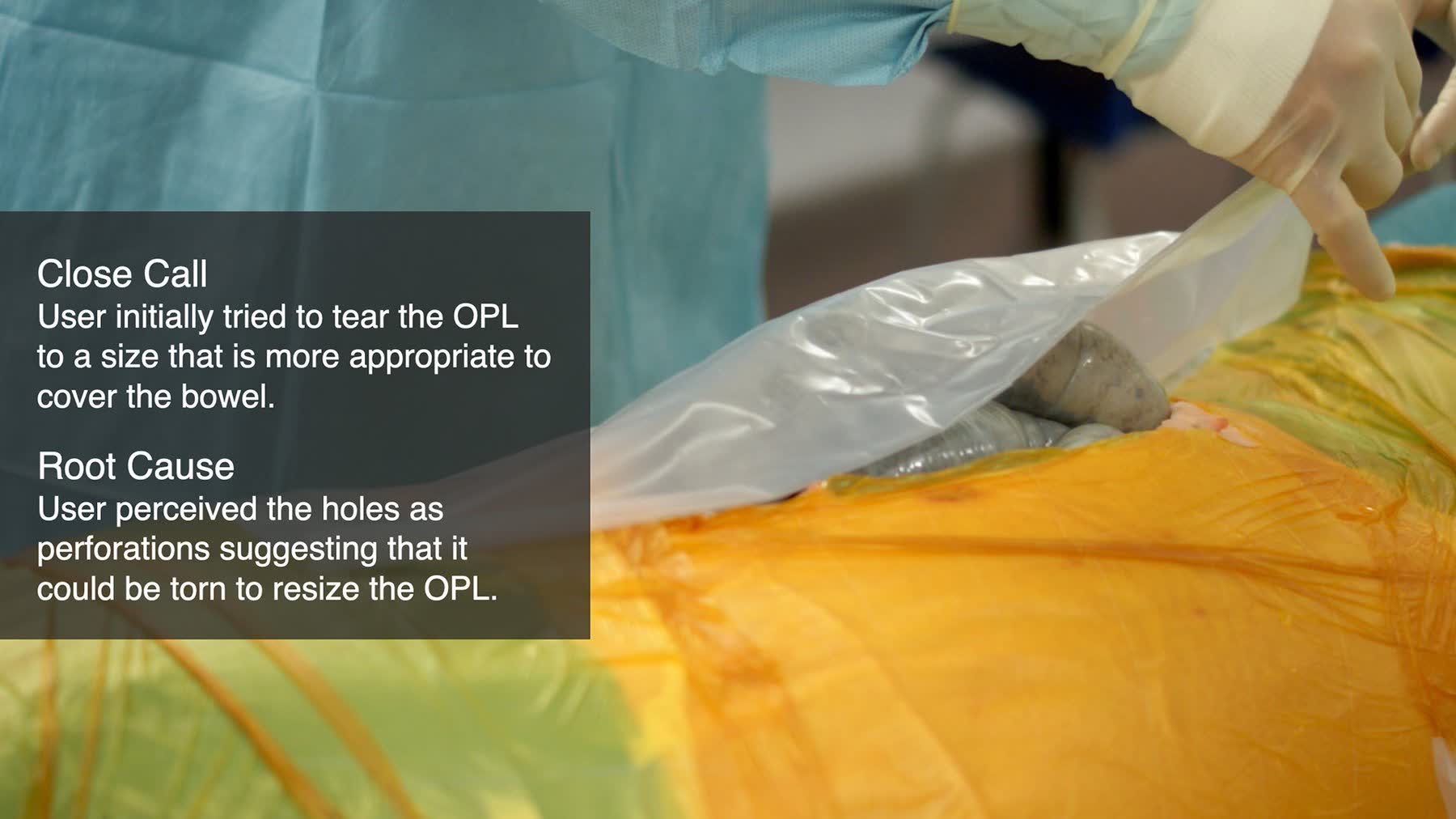
Procedural cues conflicted with surgeons' mental models
Existing instructional and physical cues within the device created cognitive interference — increasing load at exactly the moments requiring precision and confidence.
Critical steps were insufficiently differentiated
Key procedural steps were not meaningfully distinct from non-critical ones — increasing the risk of misuse through perceived equivalence rather than user error alone.
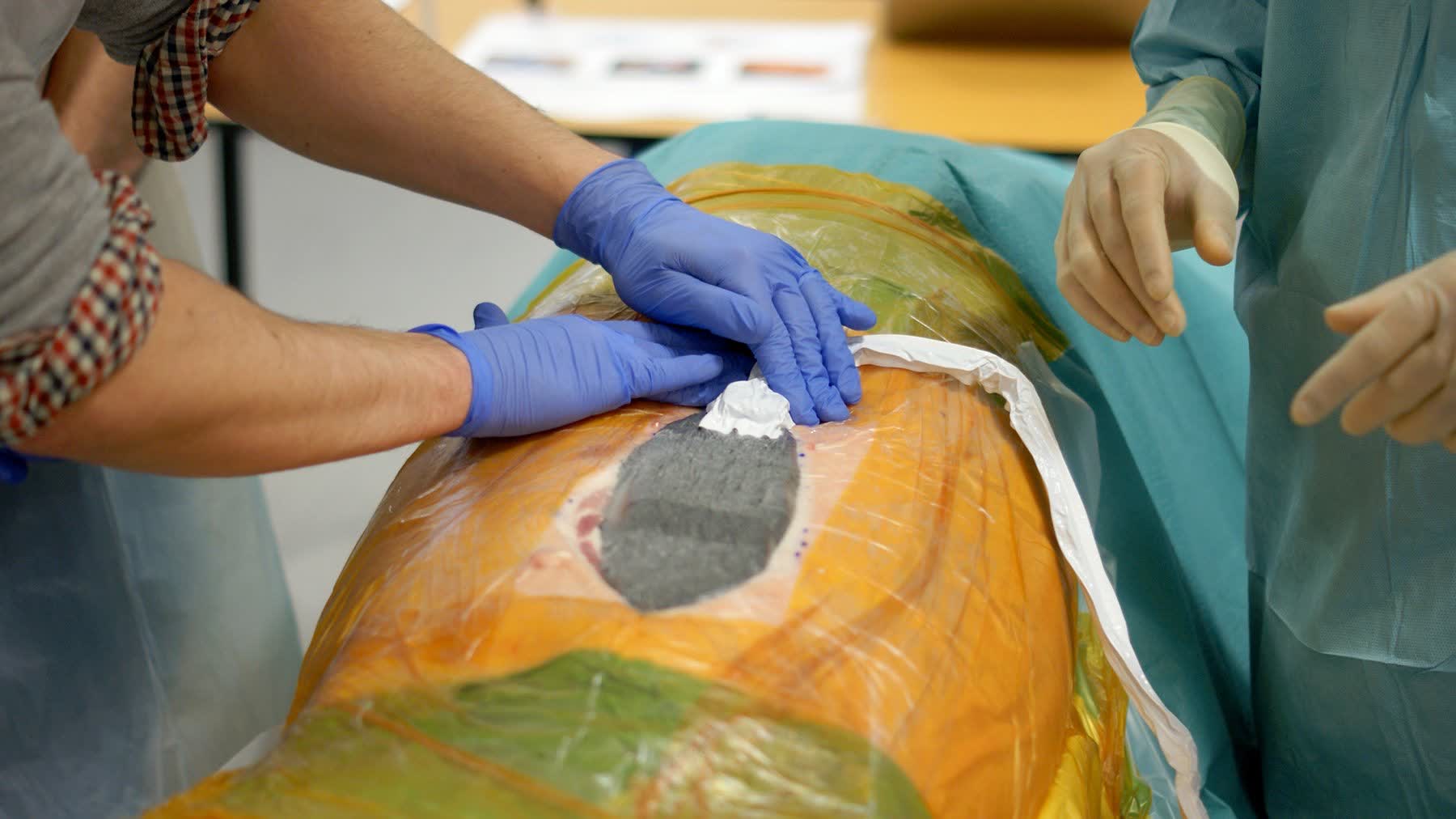
Visibility and tactile feedback outweighed speed
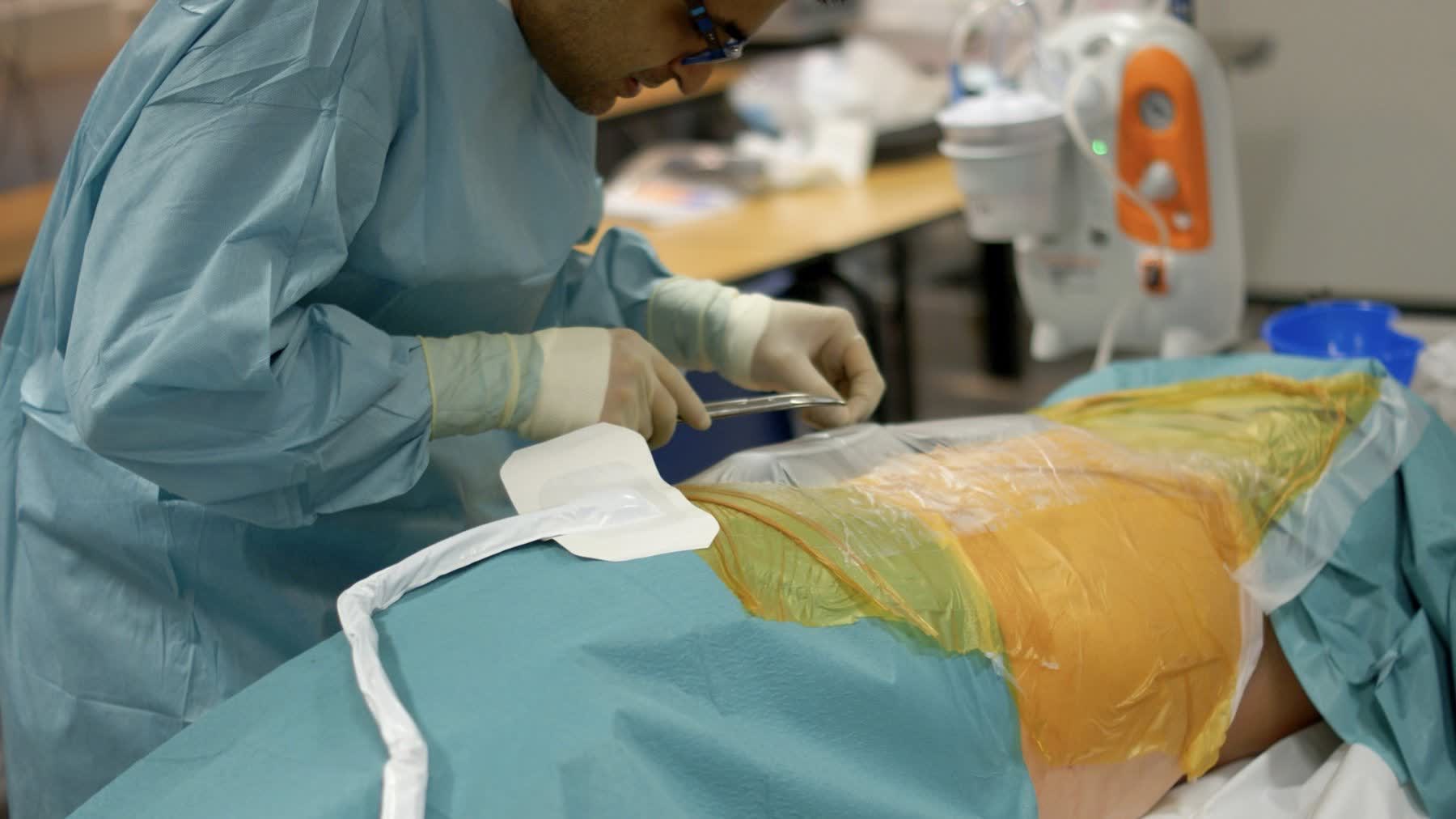
Surgeons prioritised feedback — seeing and feeling that each step had completed correctly — over operational efficiency, making haptic and visual confirmation a primary design requirement.
Approach
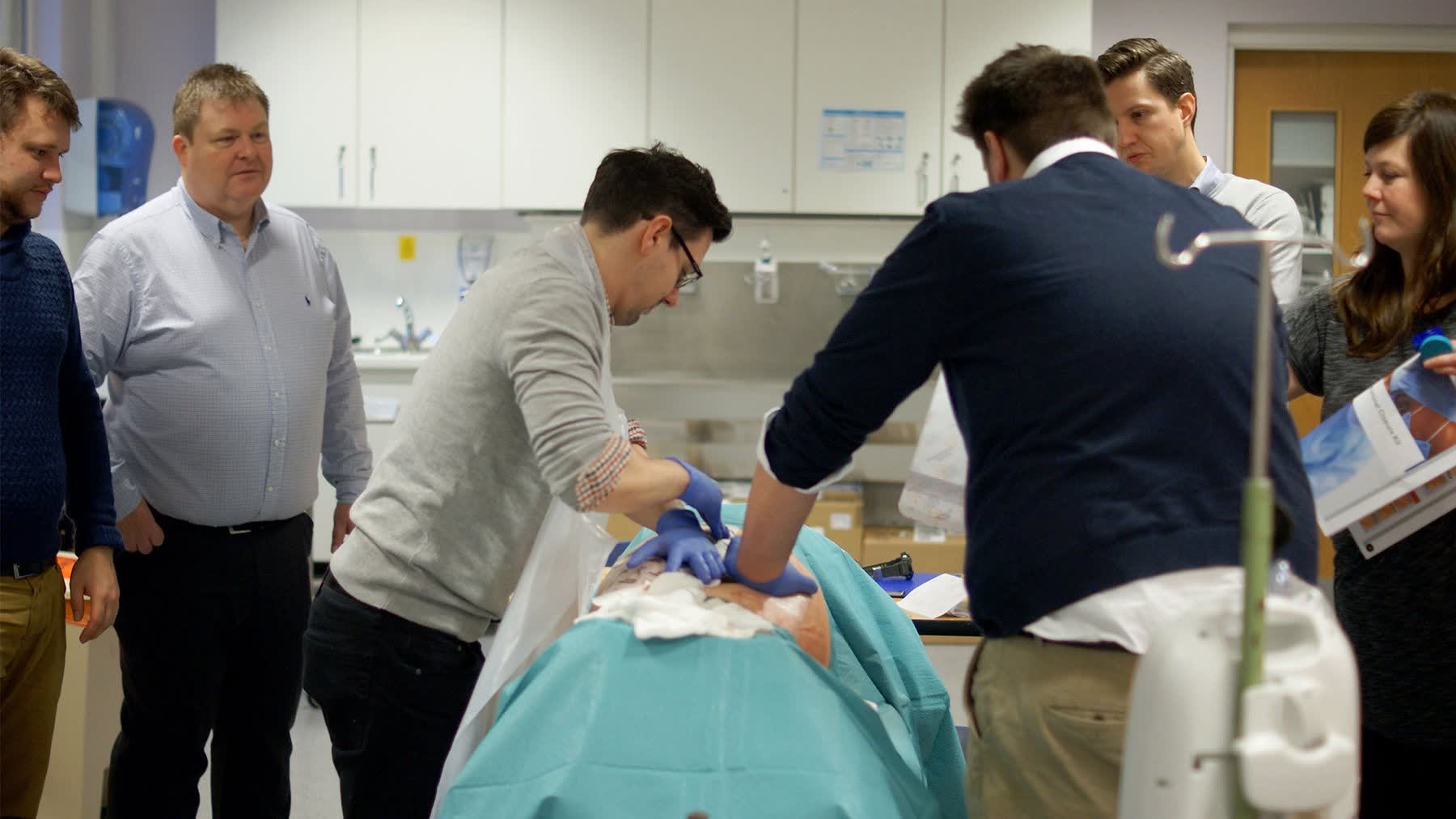
Research designed to identify safety risks and usability breakdowns in conditions that closely replicated real surgical use — preserving clinical validity throughout.
Formative usability testing
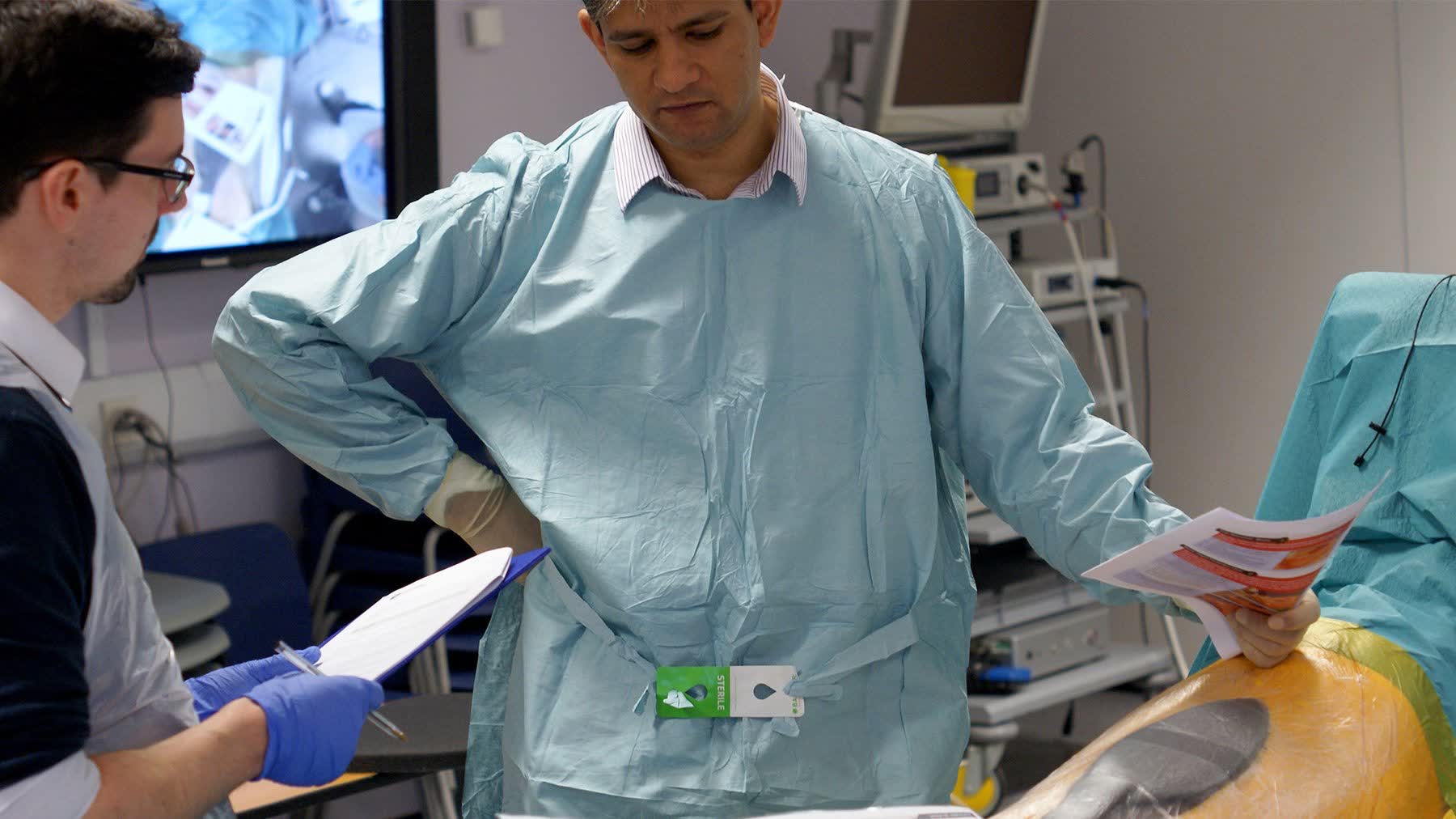
Exploratory testing in simulated surgical environments with surgeons of varying experience levels to surface usability breakdowns in realistic conditions — not abstracted lab settings.
Think-aloud protocols
Structured verbal protocols to surface decision-making under pressure — revealing how surgeons interpreted cues, resolved uncertainty, and recovered from confusion in real time.
Multi-angle video capture
Detailed interaction sequences captured from multiple angles to enable precise analysis of hand-device interaction, gaze behaviour, and procedural sequencing post-session.
Role & Ownership
Design Researcher
- Led recruitment, study design, facilitation, and synthesis across all research sessions — ensuring clinical realism was maintained throughout without compromising participant safety.
- Delivered evidence-based findings and prioritised recommendations to support safe iteration of the concept system.
- Translated complex interaction-level observations into clear, actionable guidance for the engineering and design teams responsible for the next development phase.
System & Capabilities Delivered
Evidence and guidance designed to reduce development risk and support safer design decisions before further investment.
Usability risk assessment
Prioritised catalogue of identified risks, categorised by severity, frequency, and consequence — giving the development team a clear picture of what to address first.
Human factors recommendations
Actionable design guidance linked directly to observed usability breakdowns — structured to be implementable by both design and engineering teams.
Concept refinement guidance
Strategic direction for the next phase of concept development, aligned with real-world surgical workflows and regulatory expectations for formative usability evidence.
Outcome & Impact
Reducing development risk before further investment in a safety-critical device
Critical usability risks identified before further development
Prevented unsafe design decisions from being carried forward into a more advanced and expensive stage of development.
Safer design direction aligned with surgical workflows
Recommendations were directly grounded in observed surgeon behaviour — not assumptions — ensuring they were feasible and clinically appropriate.
Supported client decision-making on next-phase development
Gave the Smith & Nephew team clear evidence to inform investment, prioritisation, and concept direction decisions for the next development phase.